Ferricyanide
 | |
 | |
| Names | |
|---|---|
| IUPAC name iron(3+) hexacyanide | |
| Systematic IUPAC name hexacyanidoferrate(III) | |
| Other names ferric hexacyanide; hexacyanidoferrate(3−); hexacyanoferrate(III) | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider |
|
| KEGG |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | [Fe(CN)6]3− |
| Related compounds | |
Other cations | Hexacyanonickelate(III) |
Related compounds | Ferrocyanide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  Y verify (what is Y verify (what is  Y Y N ?) N ?) Infobox references | |
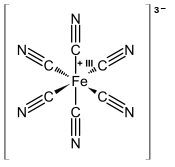
Ferricyanide is the anion [Fe(CN)6]3−. It is also called hexacyanoferrate(III) and in rare, but systematic nomenclature, hexacyanidoferrate(III). The most common salt of this anion is potassium ferricyanide, a red crystalline material that is used as an oxidant in organic chemistry.[1]
Properties
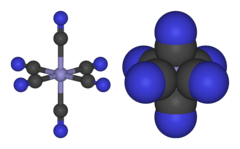
[Fe(CN)6]3− consists of a Fe3+ center bound in octahedral geometry to six cyanide ligands. The complex has Oh symmetry. The iron is low spin and easily reduced to the related ferrocyanide ion [Fe(CN)6]4−, which is a ferrous (Fe2+) derivative. This redox couple is reversible and entails no making or breaking of Fe–C bonds:
- [Fe(CN)6]3− + e− ⇌ [Fe(CN)6]4−
This redox couple is a standard in electrochemistry.
Compared to main group cyanides like potassium cyanide, ferricyanides are much less toxic because of the strong bond between the cyanide ion (CN−) and the Fe3+. They do react with mineral acids, however, to release highly toxic hydrogen cyanide gas.
Uses
Treatment of ferricyanide with iron(II) salts affords the brilliant, long-lasting pigment Prussian blue, the traditional color of blueprints.
See also
References
- ^ Gail, E.; Gos, S.; Kulzer, R.; Lorösch, J.; Rubo, A.; Sauer, M.; Kellens, R.; Reddy, J.; Steier, N.; Hasenpusch, W. (October 2011). "Cyano Compounds, Inorganic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a08_159.pub3. ISBN 978-3527306732.
- v
- t
- e
| HCN | He | ||||||||||||||||||||
| LiCN | Be(CN)2 | B(CN)3 | C(CN)4 C2(CN)2 | NH4CN ONCN O2NCN N3CN | OCN− -NCO O(CN)2 | FCN | Ne | ||||||||||||||
| NaCN | Mg(CN)2 | Al(CN)3 | Si(CN)4 (CH3)3SiCN | P(CN)3 | SCN− -NCS (SCN)2 S(CN)2 | ClCN | Ar | ||||||||||||||
| KCN | Ca(CN)2 | Sc(CN)3 | Ti | V | Cr(CN)63− | Mn | Fe(CN)2 Fe(CN)64− Fe(CN)63− | Co(CN)2 Co(CN)3− 5 | Ni(CN)2 Ni(CN)42− Ni(CN)44− | CuCN | Zn(CN)2 | Ga(CN)3 | Ge(CN)2 Ge(CN)4 | As(CN)3 (CH3)2AsCN (C6H5)2AsCN | SeCN− (SeCN)2 Se(CN)2 | BrCN | Kr | ||||
| RbCN | Sr(CN)2 | Y(CN)3 | Zr | Nb | Mo(CN)84− | Tc | Ru | Rh | Pd(CN)2 | AgCN | Cd(CN)2 | In(CN)3 | Sn(CN)2 | Sb(CN)3 | Te(CN)2 Te(CN)4 | ICN | Xe | ||||
| CsCN | Ba(CN)2 | * | Lu(CN)3 | Hf | Ta | W(CN)84− | Re | Os | Ir | Pt(CN)42- Pt(CN)64- | AuCN Au(CN)2- | Hg2(CN)2 Hg(CN)2 | TlCN | Pb(CN)2 | Bi(CN)3 | Po | At | Rn | |||
| Fr | Ra | ** | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||
| * | La(CN)3 | Ce(CN)3 Ce(CN)4 | Pr(CN)3 | Nd | Pm | Sm(CN)3 | Eu(CN)3 | Gd(CN)3 | Tb | Dy(CN)3 | Ho(CN)3 | Er | Tm | Yb(CN)3 | |||||||
| ** | Ac(CN)3 | Th(CN)4 | Pa | UO2(CN)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | |||||||










