Leptosidin
 | |
| Names | |
|---|---|
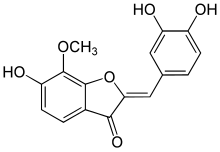
| IUPAC name 3′,4′,6-Trihydroxy-7-methoxyaurone | |
| Systematic IUPAC name (2Z)-2-[(3,4-Dihydroxyphenyl)methylidene]-6-hydroxy-7-methoxy-1-benzofuran-3(2H)-one | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
| KEGG |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C16H12O6 |
| Molar mass | 300.266 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Leptosidin was the first aurone to be isolated in Coreopsis grandiflora by Geissman T.A. and Heaton C.D. in 1943.[1] Leptosidin blocks the active residues of PRKACA.[2]
References
- ^ Leptosidin on metabolomics.jp
- ^ S, Sandeep; V, Priyadarshini; D, Pradhan; M, Munikumar; A, Umamaheswari (2012). "Docking and molecular dynamics simulations studies of human protein kinase catalytic subunit alpha with antagonist". Journal of Clinical and Scientific Research: 15–23. doi:10.15380/2277-5706.JCSR.12.005.
- v
- t
- e
Aurones and their glycosides
- 4,5,6-trihydroxyaurone
- 6,3',4',5'-tetrahydroxyaurone
- Aureusidin
- Bracteatin
- Hispidol
- Leptosidin
- Maritimetin
- Sulfuretin
- Maritimein
- Subulin
 | This article about an aromatic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











