Nitrogen triiodide
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names Nitrogen triiodide[1] Triiodoazane[1] Triiodidonitrogen[1] | |||
| Other names Nitrogen iodide Ammonia triiodide Touch Powder Triiodine nitride Triiodine mononitride Triiodamine[citation needed] Triiodoamine[citation needed] Iodine nitride | |||
| Identifiers | |||
CAS Number |
| ||
3D model (JSmol) |
| ||
| ChemSpider |
| ||
PubChem CID |
| ||
CompTox Dashboard (EPA) |
| ||
InChI
| |||
| |||
| Properties | |||
Chemical formula | NI3 | ||
| Molar mass | 394.719 g/mol | ||
| Appearance | dark solid | ||
| Boiling point | sublimes at −20 °C | ||
Solubility in water | Insoluble | ||
| Solubility | organic solvents,[2] such as diethyl ether | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards | Extremely explosive and unstable | ||
| NFPA 704 (fire diamond) |  3 0 4 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  Y verify (what is Y verify (what is  Y Y N ?) N ?) Infobox references | |||
Nitrogen triiodide is an inorganic compound with the formula NI3. It is an extremely sensitive contact explosive: small quantities explode with a loud, sharp snap when touched even lightly, releasing a purple cloud of iodine vapor; it can even be detonated by alpha radiation. NI3 has a complex structural chemistry that is difficult to study because of the instability of the derivatives.
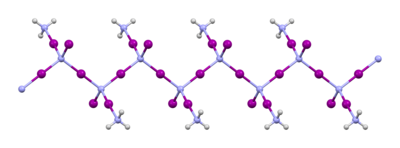
Structure of NI3 and its derivatives
Nitrogen triiodide was first characterized by Raman spectroscopy in 1990 when it was prepared by an ammonia-free route. Boron nitride reacts with iodine monofluoride in trichlorofluoromethane at −30 °C to produce pure NI3 in low yield:[3]
- BN + 3 IF → NI3 + BF3
NI3 is pyramidal (C3v molecular symmetry), as are the other nitrogen trihalides and ammonia.[4]
The material that is usually called "nitrogen triiodide" is prepared by the reaction of iodine with ammonia. When this reaction is conducted at low temperatures in anhydrous ammonia, the initial product is NI3 · (NH3)5, but this material loses some ammonia upon warming to give the 1:1 adduct NI3 · NH3. This adduct was first reported by Bernard Courtois in 1812, and its formula was finally determined in 1905 by Oswald Silberrad.[5] Its solid state structure consists of chains of -NI2-I-NI2-I-NI2-I-.[6] Ammonia molecules are situated between the chains. When kept cold in the dark and damp with ammonia, NI3 · NH3 is stable.
Decomposition and explosiveness
The instability of NI3 and NI3 · NH3 can be attributed to the large steric strain caused by the three large iodine atoms being held in proximity to each other around the relatively tiny nitrogen atom. This results in a very low activation energy for its decomposition, a reaction made even more favorable due to the great stability of N2. Nitrogen triiodide has no practical commercial value due to its extreme shock sensitivity, making it impossible to store, transport, and utilize for controlled explosions. Whereas pure nitroglycerin is powerful and also greatly shock-sensitive (although not nearly as much so as nitrogen triiodide, which can be set off with the touch of a feather), it was only due to phlegmatizers that nitroglycerin's shock sensitivity was reduced and it became safer to handle and transport in the form of dynamite.
The decomposition of NI3 proceeds as follows to give nitrogen gas and iodine:
- 2 NI3 (s) → N2 (g) + 3 I2 (g) (−290 kJ/mol)
However, the dry material is a contact explosive, decomposing approximately as follows:[4]
- 8 NI3 · NH3 → 5 N2 + 6 NH4I + 9 I2
Consistent with this equation, these explosions leave orange-to-purple stains of iodine, which can be removed with sodium thiosulfate solution. An alternate method of stain removal is to simply allow the iodine time to sublime. Small amounts of nitrogen triiodide are sometimes synthesized as a demonstration to high school chemistry students or as an act of "chemical magic."[7] To highlight the sensitivity of the compound, it is usually detonated by touching it with a feather, but even the slightest air current, laser light, or other movement can cause detonation. Nitrogen triiodide is also notable for being the only known chemical explosive that detonates when exposed to alpha particles and nuclear fission products.[8]
References
- ^ a b c per analogiam, see NF3 names, IUPAC Red Book 2005, p. 314
- ^ 4. Analytical techniques. acornusers.org
- ^ Tornieporth-Oetting, I.; Klapötke, T. (1990). "Nitrogen Triiodide". Angewandte Chemie International Edition. 29 (6): 677–679. doi:10.1002/anie.199006771.
- ^ a b Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 0-12-352651-5.
- ^ Silberrad, O. (1905). "The Constitution of Nitrogen Triiodide". Journal of the Chemical Society, Transactions. 87: 55–66. doi:10.1039/CT9058700055.
- ^ Hart, H.; Bärnighausen, H.; Jander, J. (1968). "Die Kristallstruktur von Stickstofftrijodid‐1‐Ammoniak NJ3 · NH3". Z. Anorg. Allg. Chem. 357 (4–6): 225–237. doi:10.1002/zaac.19683570410.
- ^ Ford, L. A.; Grundmeier, E. W. (1993). Chemical Magic. Dover. p. 76. ISBN 0-486-67628-5.
- ^ Bowden, F. P. (1958). "Initiation of Explosion by Neutrons, α-Particles, and Fission Products". Proceedings of the Royal Society of London A. 246 (1245): 216–219. Bibcode:1958RSPSA.246..216B. doi:10.1098/rspa.1958.0123. S2CID 137728239.
External links
- See the explosion
- Nitrogen Tri-Iodide – explains why the compound is explosive
- Nitrogen Tri-Iodide Detonation on Youtube
- v
- t
- e
| HI +H | He | |||||||||||||||||
| LiI | BeI2 | BI3 +BO3 | CI4 +C | NI3 NH4I +N | I2O4 I2O5 I2O6 I4O9 | IF IF3 IF5 IF7 | Ne | |||||||||||
| NaI | MgI2 | AlI AlI3 | SiI4 | PI3 P2I4 +P PI5 | S2I2 | ICl ICl3 | Ar | |||||||||||
| KI | CaI2 | ScI3 | TiI2 TiI3 TiI4 | VI2 VI3 | CrI2 CrI3 CrI4 | MnI2 | FeI2 FeI3 | CoI2 | NiI2 -Ni | CuI | ZnI2 | GaI GaI3 | GeI2 GeI4 +Ge | AsI3 As2I4 +As | Se | IBr IBr3 | Kr | |
| RbI RbI3 | SrI2 | YI3 | ZrI2 ZrI3 ZrI4 | NbI4 NbI5 | MoI2 MoI3 | TcI3 | RuI3 | RhI3 | PdI2 | AgI | CdI2 | InI InI3 | SnI2 SnI4 | SbI3 +Sb | TeI4 +Te | I− I− 3 | Xe | |
| CsI CsI3 | BaI2 | LuI3 | HfI3 HfI4 | TaI4 TaI5 | WI2 WI3 WI4 | ReI3 ReI 4 | OsI OsI2 OsI3 | IrI3 IrI 4 | PtI2 PtI4 | AuI AuI3 | Hg2I2 HgI2 | TlI TlI3 | PbI2 | BiI3 | PoI2 PoI4 | AtI | Rn | |
| Fr | RaI2 | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | ||||||||||||||||||
| LaI2 LaI3 | CeI2 CeI3 | PrI2 PrI3 | NdI2 NdI3 | PmI3 | SmI2 SmI3 | EuI2 EuI3 | GdI2 GdI3 | TbI3 | DyI2 DyI 3 | HoI3 | ErI3 | TmI2 TmI3 | YbI2 YbI3 | |||||
| AcI3 | ThI2 ThI3 ThI4 | PaI4 PaI5 | UI3 UI4 | NpI3 | PuI3 | AmI2 AmI3 | CmI3 | BkI 3 | CfI 2 CfI 3 | EsI2 EsI3 | Fm | Md | No | |||||












