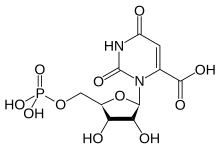
Orotidine 5'-monophosphate
 | |
| Names | |
|---|---|
| IUPAC name 2,6-Dioxo-3-(5-O-phosphono-β-D-ribofuranosyl)-1,2,3,6-tetrahydropyrimidine-4-carboxylic acid | |
| Systematic IUPAC name 3-{(2R,3R,4S,5R)-3,4-Dihydroxy-6-[(phosphonooxy)methyl]oxolan-2-yl}-2,6-dioxo-1,2,3,6-tetrahydropyrimidine-4-carboxylic acid | |
| Other names Orotidylic acid, OMP | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
| MeSH | Orotidine+5'-monophosphate |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C10H13N2O11P |
| Molar mass | 368.191 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  N verify (what is N verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
Orotidine 5'-monophosphate (OMP), also known as orotidylic acid, is a pyrimidine nucleotide[1] which is the last intermediate in the biosynthesis of uridine monophosphate.[2] OMP is formed from orotate and phosphoribosyl pyrophosphate by the enzyme orotate phosphoribosyltransferase.
In humans, the enzyme UMP synthase converts OMP into uridine 5'- monophosphate.[2] If UMP synthase is defective, orotic aciduria can result.[2]
References
- ^ PubChem. "Orotidine-5'-monophosphate". pubchem.ncbi.nlm.nih.gov. Retrieved 2023-01-26.
- ^ a b c "UMPS uridine monophosphate synthetase [Homo sapiens (human)] - Gene - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2023-01-26.
- v
- t
- e
metabolism
| anabolism |
| ||||||
|---|---|---|---|---|---|---|---|
| catabolism |
metabolism
| anabolism |
| ||||
|---|---|---|---|---|---|
| catabolism |
|
 | This biochemistry article is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e











